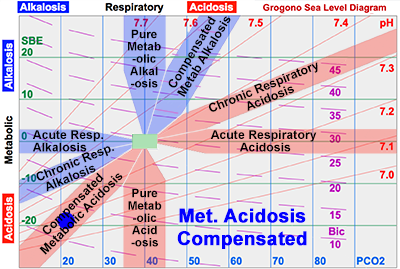
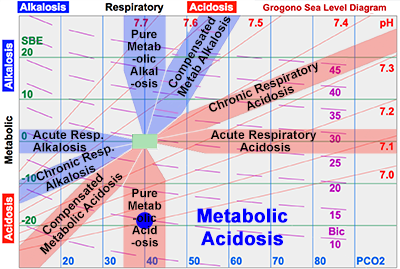
Metabolic Acidosis.
The best measure of overall metabolic acidosis at Sea Level is Standard Base Excess (SBE) and at High Altitude is Altitude Base Excess (ABE). The severity of Metabolic Acidosis appears to be the best predictor of outcome in severe trauma or following major surgery: the greater the acidity, the higher the mortality.1,2,3.
This page is a brief summary but additional information is available at: E-Medicine (Antonia Quinn, DO) or Wikipedia
Causes: The many causes of primary metabolic acidosis are commonly classified according to the anion gap:
-
Metabolic Acidosis with a Normal Anion Gap:
- Longstanding diarrhea (bicarbonate loss)
- Uretero-sigmoidostomy
- Pancreatic fistula
- Renal Tubular Acidosis
- Intoxication, e.g., ammonium chloride, acetazolamide, bile acid sequestrants
- Renal failure
-
Metabolic Acidosis with an Elevated Anion Gap:
- lactic acidosis
- ketoacidosis
- chronic renal failure (accumulation of sulfates, phosphates, uric acid)
- intoxication, e.g., salicylates, ethanol, methanol, formaldehyde, ethylene glycol, paraldehyde, INH, toluene, sulfates, metformin.
- rhabdomyolysis
Treating Severe Metabolic Acidosis.
The best treatment for a metabolic acidosis is to correct the underlying cause. In an emergency, treatment is based on the condition of the patient, supported by laboratory findings. The best measure of the level of metabolic acidosis is the Standard Base Excess (SBE) because it is independent of the PCO2. If it is decided to administer bicarbonate, the SBE and the size of the treatable space are used to calculate the dose required:
Metabolic Alkalosis
Causes: Primary Metabolic alkalosis may be caused be:
- Loss of acid via the urine, stools, or vomiting
- Transfer of hydrogen ions into the cells
- Excessive bicarbonate administration, e.g. alkali given to patients with renal failure.
- Contraction of the extracellular space due to excessive diuretic treatment
Prolonged Metabolic Alkalosis may be caused by a number of different mechanisms:
- Decrease in renal perfusion: occurs in dehydration, cardiac failure, or cirrhosis, stimulates the renin-angiotensin system which increases sodium reabsorption in the nephron.
- Chloride Depletion: may occur via vomiting or through the use of loop diuretics and this enhances bicarbonate reabsorption with associated hydrogen ion loss.
- Hypokalemia: Metabolic alkalosis may be associated with hypokalemia which can then maintain metabolic alkalosis by various mechanisms:
- Shift of hydrogen ions intracellularly which enhances bicarbonate reabsorption in the collecting duct.
- Stimulation of the H+/K+ ATPase in the collecting duct: this leads to potassium ion reabsorption and hydrogen ion secretion. The net gain of bicarbonate maintains the metabolic alkalosis.
- Renal ammonia genesis: Ammonium ions (NH4+) are produced in the proximal tubule from glutamine metabolism. Alpha-ketoglutarate is produced. The metabolism generates bicarbonate.
- Impaired chloride ion reabsorption in the distal nephron increases luminal electronegativity with enhanced hydrogen ion secretion.
- Lowered glomerular filtration rate (GFR). Hypokalemia may decrease GFR, which in turn decreases the filtered load of bicarbonate. In volume depletion this impairs excretion of the excess bicarbonate.
Treating Severe Metabolic Alkalosis
Physiological Response: Proper hydration normally allows the kidneys to correct metabolic alkalosis. However, in severe cases accompanied by hypokalemia, correction of the hypokalemia may be necessary first.
As with metabolic acidosis, it is best to treat the underlying abnormality. More active intervention is occasionally required and various techniques are available. A common transient cause is the result of therapy: when sodium bicarbonate is given to correct acute metabolic acidosis, it may leave a residual metabolic alkalosis. Time, hydration, and renal function should gradually correct this.
Contraction alkalosis is one of the easier causes to understand and treat. Dehydration concentrates the body’s electrolytes. As the extracellular fluid (pH = 7.4) is on the alkaline side of neutral (pH = 6.8), the relative alkaline mixture of electrolytes is concentrated and shifts the pH to more alkaline value. Rehydration, e.g., with oral fluids or intravenous Ringer’s lactate, restores the normal electrolyte concentration and, therefore, the pH.
Other therapies: Intravenous dilute hydrochloric acid is occasionally used but carries the risk of hemolysis. Potassium chloride may also be used unless there is kidney failure. In severe cases which are unresponsive to other measures ammonium chloride may be given (1 to 2 g orally every 4 to 6 hours up to 4 g every 2 hours. It may also be given by intravenous infusion (100 to 200 mEq dissolved in 500 to 1000 ml of isotonic saline) in addition to potassium replacement. In severe unresponsive metabolic alkalosis it may be necessary to administer hydrochloric acid or institute peritoneal dialysis. Specific therapy depends on the underlying pathology. For details visit: E-Medicine (Antonia Quinn, DO).
Urgent Metabolic Therapy – Calculating the Dose
The Body’s Metabolism produces respiratory (carbonic) acid and, in ischemia or cardiorespiratory failure, metabolic (lactic) acid. Urgent therapy for a metabolic problem is usually required for metabolic acidosis.
Reasons to Limit Bicarbonate Dose.
1. Causes Respiratory Acidosis.
The Injected Bicarbonate initially enters the three-liter plasma volume instead of the calculated 21 liters of treatable volume (it does not cross the cell membrane to enter the two liters of red cells). This transiently “over-treats” this small compartment.
Worse, the bicarbonate ions combine with hydrogen ions to form CO2. The raised the PCO2 level initially causes a respiratory acidosis. For each 100 mEq of bicarbonate converted, about 2.24 liters of carbon dioxide have to be exhaled, equivalent to ten minutes normal production. It is customary to administer a half dose to correct the metabolic disturbance half way; continuing ventilation eliminates this excess CO2 to reach a more modest level of partially compensated metabolic acidosis.
Bicarbonate Therapy:
The intention, of course, is to normalize the “Bath Water”, – the extracellular fluid – (20% of the body or about 14 liters). But, because the injected bicarbonate also equilibrates to some extent with the intracellular fluid, the “treatable volume” is larger, say 30% or 21 liters.
Full Dose: The diagram shows an example of a patient with a (pure) metabolic acidosis, SBE = -18 mEq/L. To achieve complete correction for someone weighing 70 kg:
0.3 x Wt (kg) x SBE (mEq/L) = Dose (mEq)
0.3 x 70 x 18 = 378 mEq
Half Dose: It is customary to give about half this dose – which returns the metabolic disturbance about half way back towards normal, i.e., about half way to the zero line. In this case 189 mEq corrects the SBE to -9 mEq/L. There are several other good reasons to avoid administering large doses of bicarbonate:
2. Initially Raises Intracellular PCO2
The Carbon Dioxide Produced enters the cells freely, unlike the bicarbonate ions which have been administered. This initially causes the intracellular fluid to become more acid. However, direct studies employing nuclear magnetic resonance indicate that this change may be insignificant (Severinghaus, Personal Communication 1986).
3. Subsequent Residual Changes.
The Residual Effects of bicarbonate are all interrelated. Nevertheless, it is helpful to consider them separately as it emphasizes the importance of caution when considering administering bicarbonate.
![]() Sodium Ions: The injected bicarbonate ions are accompanied by sodium ions which will therefore be responsible for a subsequent raised sodium level – a hypernatremia.
Sodium Ions: The injected bicarbonate ions are accompanied by sodium ions which will therefore be responsible for a subsequent raised sodium level – a hypernatremia.
![]() Osmolarity: The hypernatremia will increase the osmolarity of the extracellular fluid. In combination with other therapy, such as intravenous glucose, this hyperosmolarity may be critical and even cause coma. In neonates, rapid infusion of bicarbonate may cause intracranial hemorrhage.
Osmolarity: The hypernatremia will increase the osmolarity of the extracellular fluid. In combination with other therapy, such as intravenous glucose, this hyperosmolarity may be critical and even cause coma. In neonates, rapid infusion of bicarbonate may cause intracranial hemorrhage.
![]() Metabolic Alkalosis: Once the underlying pathology causing the metabolic acidosis is corrected, then the bicarbonate therapy will be responsible for a residual iatrogenic metabolic alkalosis.
Metabolic Alkalosis: Once the underlying pathology causing the metabolic acidosis is corrected, then the bicarbonate therapy will be responsible for a residual iatrogenic metabolic alkalosis.
![]() Caution: For all of the above reasons, bicarbonate therapy tends to be reserved for emergencies and situations where indications for therapy are compelling.
Caution: For all of the above reasons, bicarbonate therapy tends to be reserved for emergencies and situations where indications for therapy are compelling.
References:
- Gunnerson KJ, Saul M, He S & Kellum JA:
Lactate versus non-lactate metabolic acidosis: a retrospective outcome evaluation of critically ill https://ccforum.biomedcentral.com/articles/10.1186/cc3987 - Wernly B, Heramvand N, Masyuk M, Rezar R, Bruno RR, Kelm M, Niederseer D, Lichtenauer M, Hoppe UC, Bakker J, & Jung C:
Acidosis predicts mortality independently from hyperlactatemia in patients with sepsis. European Journal of Internal Medicine, 76: 76-81, June 2020 - Zante BJ, Reichenspurner H, Kubik M, Kluge S, Schefold JC, Pfortmueller CA:
Base excess is superior to lactate-levels in prediction of ICU mortality after cardiac surgery. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0205309