Anonymous
Page Index.
- Anion Gap
- Effects of temperature.
- Kilopascals for PCO2
- Converting pressures in different units
- Arterial vs. End-exhaled PCO2 difference
- Summation of PO2 and PCO2
- Therapeutic implications
This page covers selected clinical topics. Separate pages deal with the treatment of Respiratory and Metabolic disturbances. In addition, the Rapidity of Compensation is covered in the page on the production of acid.
Anion Gap.
Some causes of metabolic acidosis, e.g., lactic acidosis, release anions into the extracellular fluid which may not be measured. When this occurs there will be an unexpected discrepancy between the sums of the principal cations and anions. The usual sum is:
Gap = Na+ + K+ – Cl– – HCO3–
15 = 140 + 5 – 105 – 25 mMol/L
The “gap” may be increased by other unmeasured anions, e.g., lactate, phosphate, or sulfate. A gap greater than 30 indicates a significant concentration of unmeasured anions. This calculation depends upon the accuracy of the other measurements; small errors, therefore, disproportionately affect in the “gap”. When information is required about these anions, it is best to measure their concentration directly, i.e., lactate in tissue hypoxia, 3-hydroxybutyrate in diabetic ketosis, and phosphate or sulfate in renal failure.
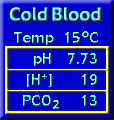
Effects of Temperature
We normally assess blood gases at 37oC. When blood is cooled, carbon dioxide becomes more soluble; this reduces the PCO2 by about 4.5% per oC. The pH rises about 0.015 per oC because hemoglobin, the principal buffer in the blood, accepts more hydrogen ions on its alpha imidazole group when cooled. However, as it is rewarmed the blood returns to normal again. Such changes typically occur when blood reaches a cold extremity (fishing in a cold stream) but is normal again when it returns to the heart. Analogous changes occur when a poikilothermic (“cold-blooded”) animal is cooled and then rewarmed. For these reasons it is best to judge the normality of human blood at 37oC. Our familiarity with the PCO2 and pH at normal body temperature makes recognition easy. So, make measurements at 37oC: normal values can be assumed to satisfactorily meet the needs of the cold tissues (Reeves and Rahn, 1979).
To Compare Partial Pressures in Air and Blood, however, it is necessary to calculate the values in the arterial blood for the patient’s actual temperature. Only then can the arterial blood gas values be meaningfully compared to exhaled gas partial pressures, or to the gases in a pump-oxygenator. Unfamiliar, however, they are useless to judge “normality”; there is no such thing as a “normal” hypothermic person to use as a standard.
Kilopascals.
Pascal and Kilopascal:
Spill a “newton” on a Square Meter: When one newton (a small cup of coffee – 102 grams weight or about 3.6 ounces of fluid) is spilled onto a one square meter table, the resulting layer of water is 0.102 mm deep. This is, by definition, the pressure exerted by one pascal. A pressure of one thousand pascals (1 kPa) is about 10.2 cm H2O or 7.75 mmHg.
One kilopascal is almost the same as the one percent of atmospheric pressure. For example, the normal arterial PCO2 of 40 mmHg is 5.33 kPa or 5.61 % of atmospheric pressure.
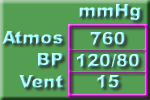
Pressure Conversion (Watch Pictures).
The pictures and tables below shows useful approximations. We should all be able to quickly convert between the various units used for the measurement of pressure. It is helpful to recognize that, for practical purposes:
760 mmHg = 1000 cmH2O = 1 atmos = 100 kPa
or, more useful to clinicians:
7.6 mmHg = 10 cmH2O = 1% atmos = 1 kPa
Difference between arterial PACO2 and end-exhaled PaCO2.
 Discrepancy
Discrepancy
Normal Health: End-expiratory PACO2 and arterial PaCO2 differ very little. During anesthesia, end-expiratory PACO2 is commonly lower than a simultaneous arterial PaCO2 due to several factors:
Body Temperature: Anesthetized patients are often cold. A blood gas analyzer routinely warms the blood to 37oC. The dissolved CO2 then exerts a higher partial pressure than it did in-vivo. This discrepancy between the low lung partial pressure and the higher blood gas analyzer partial pressure can be minimized by keeping the patient warm (!) or by requesting blood gas values calculated for the body’s actual temperature.
Lung Function: Unperfused areas of the lung behave as dead space. From such areas, the exhaled gas contains no CO2; when this gas mixes with gas from other, functioning alveoli, it reduces the partial pressure of the end-exhaled PACO2.
Rapid Ventilation: During rapid ventilation, there may be too little time for the end-tidal gas to reach its final “plateau” value. The measured value of the PACO2 may, therefore, be lower than the “true” end-tidal PACO2.
Mixing in Sample Tubing: Side-stream analyzers employ a length of sample tubing between the breathing circuit and the measuring device. In this tube, the more rapidly advancing central gas core mixes with the other gases and blunts the peak of the waveform. This tends to reduce the measured value of the PACO2.
A Check: Summation of PO2 and PCO2.
 Nitrogen and Water Vapor:
Nitrogen and Water Vapor:
The body neither consumes nor manufactures nitrogen. It might be assumed that the partial pressure of Nitrogen, the PN2, would be constant during the journey from air to tissues. However there are slight changes. During inspiration humidification occupies some of the total pressure and reduces both PN2 and PO2. The body usually consumes more O2 than the CO2 created: this concentrates the nitrogen and the PN2 rises very slightly.
However, alveolar PN2 and partial pressure of water vapor vary very little. Oxygen and carbon dioxide share the remaining space. With normal lung function and accurate analysis, the arterial blood gas values reflect this.
Normally, breathing air: PaO2 + PaCO2 = 140 mmHg
If the sum of PO2 + PCO2 is greater than expected, then the analysis contains an error. A sum which is smaller suggests that the lungs are failing to adequately transfer oxygen. For greater accuracy, this sum incorporates the respiratory quotient (RQ). Then, at atmospheric pressure breathing room air, the corrected sum of the respiratory gases is 150 mmHg:
Accurately, breathing air: PaO2 + PaCO2 / RQ = 150 mmHg
This degree of accuracy is not required for most purposes. Moreover, the RQ is usually unknown. Accordingly it is simpler and reasonable to use the first equation and add the PaCO2 and PaO2 directly:
Therapeutic implications.
 The pH is not a Disease
The pH is not a Disease
Both pH and PCO2 normally vary very little. They demonstrate predictable changes with many disease states. Understandably, the pH was itself perceived as a disease entity. However, this concept has been refuted by demonstrations by Xu et al that the body and brain tolerate pH values as low as 6.2 without damage provided circulation and oxygenation are maintained. In addition, rats in hyperbaric conditions can survive undamaged when subjected to a PCO2 exceeding one atmosphere.
An Indicator
The implication of these results is that an abnormal acid-base value is best regarded as an indicator of pathology, not as a disease in its own right.
Pharmacology
Variation in the pH alters the degree of ionization of proteins and many drugs. As most ionized substances do not cross cell membranes readily, alterations in pH affect both cellular function and the potency of many pharmaceutical agents. Relative acidity of tissues, for example in the vicinity of an abscess, is recognized to reduce the efficacy of local anesthetic solutions. Conversely, relative alkalinity enhances the uptake of local anesthetic solutions. Alkalinity also potentiates drugs such as meperidine and morphine by increasing the availability of lipophilic, uncharged base, to cross the blood-brain barrier (Shulman et al 1984).