Ernst Haas
Introduction:
Purpose. This page describes the interpretation of acid-base blood gas results using a logical approach.
Constraints of Not Knowing Patient Details:
In an Ideal World information about a patient is usually available long before the acid-base values. What follows is a logical framework for looking at acid-base values without a patient. The results invite you to “Consider” a single clinical problem. However, identical results may also be obtained from a complex combination of clinical problems and therapy.
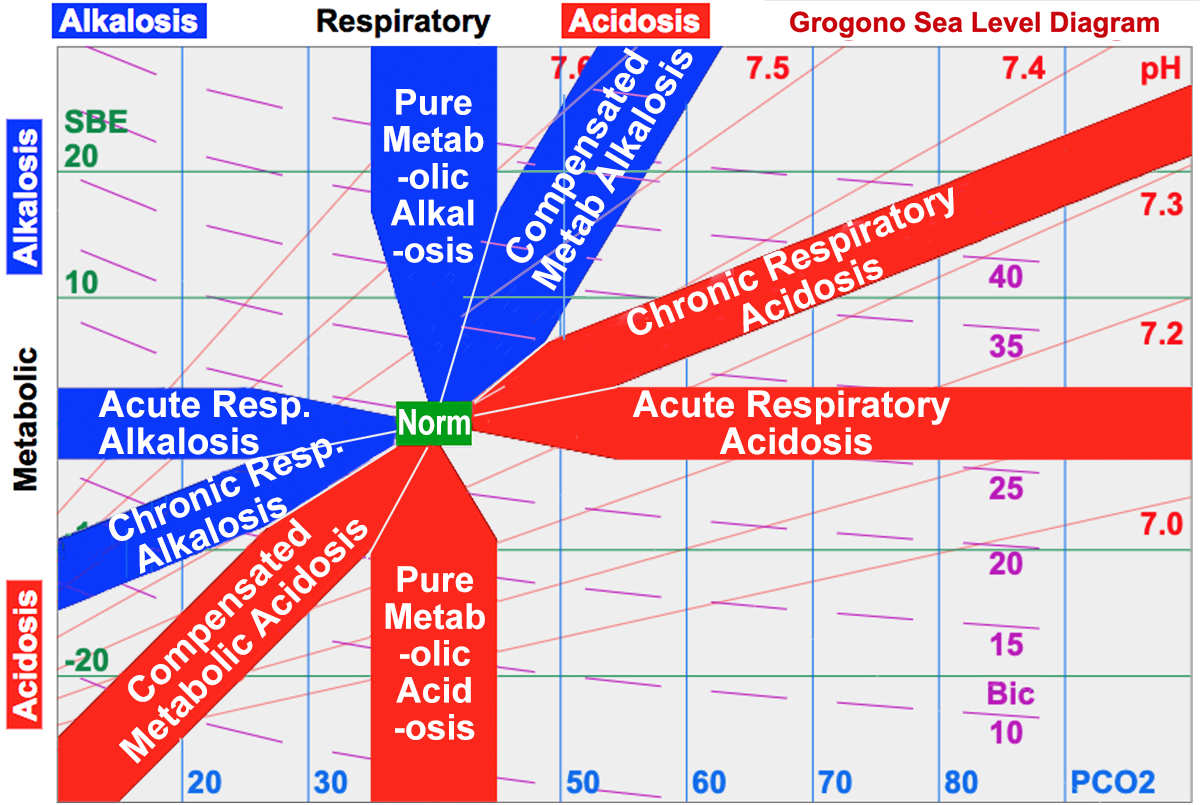
1. The pH – the Overall Change:
Is the pH normal, acid, or alkaline – critical because it governs all the subsequent thinking. In acute problems the change is usually acidic – a low pH – e.g., 7.2 or 7.1. This is because failure, both respiratory and metabolic, results in the accumulation of acids. The following paragraphs assume acidemia. However, also look at the Six Classical Disturbances below.
2. PCO2 – the Respiratory Component:
If the respiratory change is also acid (raised PCO2), then the cause is respiratory, unless the metabolic component is also acidic – in which case both are contributing to the acidic pH.
If the PCO2 is not like the pH, i.e., the PCO2 is low (alkaline), then the primary problem must be metabolic and the low PCO2 is compensating for the metabolic acidosis.
3. Standard Base Excess – the Metabolic Component:
If the Standard Base Excess (SBE) is acidic (a negative SBE), then the cause is metabolic. The exception, as above, is when the respiratory component is also acid when both are contributing to the acid pH. If the SBE is not like the pH, i.e., the SBE is alkaline, then the primary problem is not metabolic; the high SBE is a compensation for the respiratory acidosis.
4. Adjectives to Describe Changes:
Few adjectives are normally used to characterize magnitude in casual conversation. The interactive diagram necessitated a disciplined sequence: No, Minimal, Mild, Moderate, Marked, Severe. In the diagram, these adjectives are applied to both the respiratory and metabolic components and, at pH=7.4, they “balance” with the PCO2 and the SBE being equal and opposite. The steps chosen are shown in the following table:
| Adjective | PCO2 mmHg |
SBE mEq/L |
|
|---|---|---|---|
| Acidosis | Severe | > 70 | < -16.5 |
| Marked | 61 to 70 | -12 to -16.5 | |
| Moderate | 54 to 61 | -8 to -12 | |
| Mild | 48 to 54 | -4.5 to -8 | |
| Minimal | 43 to 48 | -2 to -4.5 | |
| Normal | Normal | 37 to 43 | 2 to -2 |
| Alkalosis | Minimal | 32 to 37 | 2 to 4.5 |
| Mild | 26 to 32 | 4.5 to 8 | |
| Moderate | 19 to 26 | 8 to 12 | |
| Marked | 10 to 19 | 12 to 16.5 | |
| Severe | < 10 | > 16.5 |
“Pure” Disturbances:
A pure, or acute, respiratory disturbance is found close to the SBE=0 line: the change in ventilation has occurred too rapidly for metabolic compensation to occur.
A pure metabolic disturbance would lie close to PCO2 = 40 mmHg (5.33 kPa). In practice, however, this is rare because partial respiratory compensation occurs promptly.
5. Recognizing Typical Compensated Zones:
Characteristic Zones indicate where someone with a prolonged single problem is likely to lie (see the Acid-Base Diagram). These partially compensated zones lie roughly half way between no response and complete response.
A respiratory acidosis with a PCO2 of 60 mmHg (raised by 20mmHg) would if “completely compensated” have an SBE = 12 mEq/L. An SBE=0 mEq/L would suggest “not compensated“. A value in the middle (SBE = 6 mEq/L) is typical “partially compensated chronic respiratory acidosis“.
A metabolic acidosis with SBE of -12 (reduced by 12 mEq/L) would require to be “completely compensated” a PCO2 = 20 mmHg. A normal PCO2 would indicate “not compensated“. A value in the middle (30 mmHg) is typical “compensated metabolic acidosis“.
6. Six Classical Disturbances:
The first table below summarizes the six classical acid-base disturbances to be recognized.
The second table shows four combinations that are occasionally encountered: two pure uncompensated metabolic disturbances and the two combined disturbances.
1. Six Classical Acid-Base Disturbances |
||||
| pH | PCO2 | SBE | Interpr-etation | Compensated |
|---|---|---|---|---|
| Acid | Acid | Alk | Resp Acid Comp |
SBE Half way – Normal Metabolic Comp |
| Norm | Resp Acid Pure |
SBE Normal – No Metabolic Comp | ||
| Alk | Acid | Metabolic Acid Comp | PCO2 Half way – Normal Resp Comp | |
| Alk | Alk | Acid | Resp Alk Comp |
SBE Half way – Normal Metabolic Comp |
| Norm | Resp Alk Pure |
SBE Normal – No Metabolic Comp | ||
| Acid | Alk | Metabolic Alk Comp |
PCO2 Half way – Normal Resp Comp | |
2. Four Other Acid-Base Disturbances |
||||
| pH | PCO2 | SBE | Interpr-etation | Compensated |
| Acid | Norm | Acid | Metabolic Acid Pure |
PCO2 Normal – No Resp Comp |
| Acid | Acid | Combined Acidosis | No Compensation – Both Acid | |
| Alk | Norm | Alk | Metabolic Alk Pure |
PCO2 Normal – No Resp Comp |
| Alk | Alk | Combined Alkalosis | No Compensation – Both Alkaline | |
Recognize typical zones: The red and blue zones represent the characteristic zones where patients with a single problem are typically found. The grey colored zones are rarer and therefore less important for recognition:
- Although pure metabolic acidosis and alkalosis are logically possible, people normally compensate by adjusting their ventilation.
- People with combined acidosis or alkalosis have multiple problems and do not, therefore, display a characteristic response to a single condition.
7. Examples of Interpretation:
Logical Approach when the pH is Acid:
- When the PCO2 is also acid (above 40 mmHg or 5.33 kPa) then the PCO2 contributes to the acidity.
- If not (i.e., PCO2 is alkaline) then the acidity must be metabolic and the PCO2 is compensatory.
- Is either PCO2 or SBE normal? If so, the other is a pure acidosis. Pure respiratory acidosis occurs fairly frequently, pure metabolic rarely as explained above.
- Typical Compensation lies roughly half way between no compensation and complete compensation – see The Acid-Base Diagram.
- When both components are acid, this is not a typical single condition but a combined metabolic and respiratory acidosis.
Example A: pH = 7.2, PCO2 = 60 mmHg (8 kPa), SBE = 0 mEq/L
- Overall change is acid.
- Respiratory change is also acid – therefore contributing to the acidosis.
- SBE is normal – no metabolic compensation. Therefore, pure respiratory acidosis.
- Typical of acute respiratory depression. Magnitude: marked respiratory acidosis
Example B: pH = 7.35, PCO2 = 60 mmHg (8 kPa), SBE = 6.7 mEq/L
- Overall change is slightly acid.
- Respiratory change is also acid – therefore explaining the acidosis.
- Metabolic change is alkaline – therefore compensatory.
- The respiratory acidosis is 20 mmHg (2.7 kPa) on the acid side of normal. Complete compensation would require an SBE of 11.4 mEq/L.
- The actual SBE is 6.7 eEq/L, which is roughly half way between 0 and 11.4, i.e., a typical metabolic compensation.
- Magnitude: marked respiratory acidosis with typical moderate metabolic compensation
Example C: pH = 7.15, PCO2 = 60 mmHg (8 kPa), SBE = -6 mEq/L
- Overall change is acid.
- Respiratory change is acid – therefore contributing to the acidosis.
- Metabolic change is also acid – therefore combined acidosis.
- The components are pulling in same direction – neither can be compensating for the other
- Magnitude: marked respiratory acidosis and mild metabolic acidosis
Example D: pH = 7.30, PCO2 = 30 mmHg (4 kPa), SBE = -10 mEq/L
- Overall change is acid.
- Respiratory change is alkaline – NOT therefore explaining the acidosis.
- Metabolic change is acid, i.e., responsible for the acidosis with some respiratory compensation
- The metabolic acidosis is 10 mEq/L on the acid side of normal. Complete compensation would require a fall in the PCO2 of 17 mmHg (2.3 kPa) to 23 mmHg (3.1 kPa)
- The actual PCO2 is 30 mmHg (4 kPa) which is roughly half way towards normal, i.e., a typical respiratory compensation.
- Magnitude: marked metabolic acidosis with typical mild respiratory compensation.
Conclusion
I appreciate feedback and would enjoy hearing from you and considering your suggestions. Thank you.