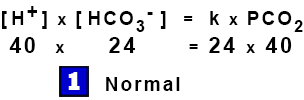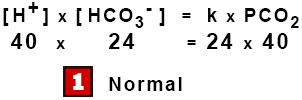Carbonic Acid (= CO2)
Carbonic acid (H2CO3) is central to our understanding and evaluation of acid-base disturbances. This is because it is so readily and rapidly changed. The dissociation products and the ionization products are normally in equilibrium:
[ H+ ] x [ HCO3– ] = k1 x H2CO3 = k2 x [ CO2 ] x [ H2O ]
This equation can be simplified because H2CO3 is not of clinical interest, [H2O] is constant in-vivo, and PCO2 is more familiar than [CO2]:
[ H+ ] x [ HCO3– ] = k x PCO2
This is the Modified Henderson Equation, an example of the Law of Mass Action where the products of the concentrations on one side are proportional to the products on the other.
Respiratory Acidosis (just CO2)
Pure Respiratory Acidosis (high PCO2) causes molecules of CO2 and water to form carbonic acid which ionizes to increase both [HCO3–] and [H+]. The [H+] changes relatively slightly due to buffering, mostly by hemoglobin. At a raised PCO2, the kidney compensates by eliminating [H+]. To maintain chemical equilibrium the [HCO3–] rises further, i.e., respiratory acidosis raises the bicarbonate level and typical partial metabolic compensation raises it further. Watch the Equation:
Metabolic Acidosis (Other Acids)
Pure Metabolic Acidosis implies a raised [H+] level with a normal PCO2. To maintain the equilibrium, the high [H+] would merely cause a reciprocal fall in the [HCO3–]. In practice typical partial respiratory compensation lowers the PCO2 almost at once, which reduces both the [H+] and the [HCO3–], i.e., metabolic acidosis lowers the bicarbonate level and typical partial respiratory compensation lowers it further. Watch the Equation:
Bicarbonate
Measures Neither!
Bicarbonate measures neither metabolic acidosis nor respiratory acidosis. This is because both the respiratory and the metabolic components affect bicarbonate ion concentration. The exception would be when Blood Chemistry show a bicarbonate change and the patient appears to have normal lungs. This usually indicates a metabolic abnormality.
Cell Membrane.
Lipid Conduit but Polar Barrier
The Cell Membrane provides a protected environment for the reactions which sustain life. It limits transfer of various substances, particularly those that are polar or ionized but allows water, lipid soluble substances, and dissolved gases to pass freely. The composition of the cell depends upon the pH for two reasons: first, as the pH changes so will the degree of ionization and, hence, the concentration of ionized substances; second, if the degree of ionization changes greatly, a substance may cross the cell membrane more or less readily.
The pH varies from one part of the cell to another and probably averages close to neutral – which is pH 6.8 at 37oC. This is more acidic than the relatively akaline extracellular fluid. In practice we neither measure, nor directly treat, the pH inside the cell; we treat the extracellular fluid.
Extracellular Fluid.
The Cell’s “Bath Water”
About 20% of the body water is extracellular fluid – typically 14 liters. This is the environment – the “Bath Water” which provides the cell’s nutrition, oxygenation, waste removal, temperature, and alkaline environment. Normal extracellular pH (7.4) is slightly alkaline and represents [H+] = 40 nmol/1. This is about 25% of the [H+] inside the cell, 160 nmol/1 (average pH = approximately 6.8). This concentration gradient favors hydrogen ion elimination from the cell but is counterbalanced by the intracellular potential of -60 mV which tends to attract the hydrogen ion into the cell.
“Treatable” Volume.
The Cell’s Larger Bath
The “Treatable Volume” is larger than the extracellular fluid because cell membranes are not completely impermeable – some equilibration occurs between the cell and the extracellular fluid. It is, therefore, customary to make calculations based on a larger volume – about 30%.
Thus the treatable space is typically about 21 liters, a useful approximation for emergency therapy. Over a longer period, however, equilibration continues between the intra- and extra- cellular fluid which further increases the apparent size of the treatable space. In addition, there may be other sources of change during a period of therapy, because the body may be either correcting the abnormality or making it worse.
Intracellular Fluid.
pH 7.0 at 37oC (alkaline !)
The Intracellular Fluid is a complex environment made up specialized regions with different functions. The pH varies from one part to another. It is commonly assumed that the intracellular pH is approximately neutral (pH 6.8 at body temperature). However, measurements by Sahlin et al (1997) indicated that the average intracellular pH may be closer to pH 7.0 with a bicarbonate concentration of about 10.2 mMol/L.
Acid Production & Elimination.
Our Fuel makes Acid
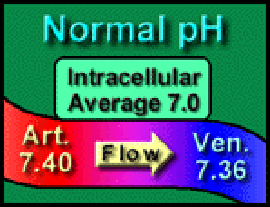 As fire makes smoke, so metabolism makes acid – CO2 and metabolic acids – which are released into the capillaries and makes the blood slightly more acidic. The blood arrives in the arterioles with a pH of 7.4 (equivalent to [H+] = 40 nMol/L) and leaves the venules with an average pH of 7.36 (equivalent to [H+] = 44 nMol/L) – as though hydrogen ions fall down a concentration gradient. Not surprisingly, acidosis is the commonest abnormality – usually partially compensated.
As fire makes smoke, so metabolism makes acid – CO2 and metabolic acids – which are released into the capillaries and makes the blood slightly more acidic. The blood arrives in the arterioles with a pH of 7.4 (equivalent to [H+] = 40 nMol/L) and leaves the venules with an average pH of 7.36 (equivalent to [H+] = 44 nMol/L) – as though hydrogen ions fall down a concentration gradient. Not surprisingly, acidosis is the commonest abnormality – usually partially compensated.
Acid Elimination.
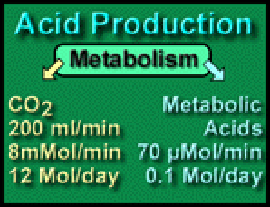
 Respiratory Acid: The quantity of CO2 produced per day is 200 milliliters per minute or (0.2 * 60 * 24) = 288 liters per day. One mole of a gas occupies 22.4 liters at STP, so approximately 12 moles of carbon dioxide are produced daily. This enormous quantity matches an appropriately powerful means of elimination – our lungs.
Respiratory Acid: The quantity of CO2 produced per day is 200 milliliters per minute or (0.2 * 60 * 24) = 288 liters per day. One mole of a gas occupies 22.4 liters at STP, so approximately 12 moles of carbon dioxide are produced daily. This enormous quantity matches an appropriately powerful means of elimination – our lungs.
Metabolic Acid. Normally only about 0.1 moles (100 mEq) of metabolic acid are produced per day – eliminated by the kidney or metabolized by the liver; appropriately their capacity is less than that of the lungs. Metabolic acids include lactic, pyruvic, and the keto-acids of diabetic acidosis.
room, therefore, an abnormal PCO2 is not usually associated with a metabolic “compensation”: there, a metabolic acidosis or alkalosis reflects a metabolic disturbance or, possibly, compensation for a long-term chronic respiratory problem.
Contrasts in Compensation.
 Rapid Respiratory Compensation. The lungs can excrete large quantities of carbon dioxide rapidly.So, unless the respiratory system is diseased or depressed, metabolic acidosis and metabolic alkalosis normally elicit prompt partial respiratory compensation.
Rapid Respiratory Compensation. The lungs can excrete large quantities of carbon dioxide rapidly.So, unless the respiratory system is diseased or depressed, metabolic acidosis and metabolic alkalosis normally elicit prompt partial respiratory compensation.
Slow Metabolic Compensation. The kidneys excrete metabolic acids more slowly; a patient may ventilate at an abnormal PCO2 for a day or two before characteristic, partial compensation develops. In the operating room or emergency room, therefore, an abnormal PCO2 is not usually associated with a metabolic “compensation”: a metabolic acidosis or alkalosis reflects a metabolic disturbance or, possibly, compensation for a long-term chronic respiratory problem.